Mesoporous titanium dioxide nanocarrier with magnetic-targeting and high loading efficiency for dual-modal imaging and photodynamic therapy†
Abstract
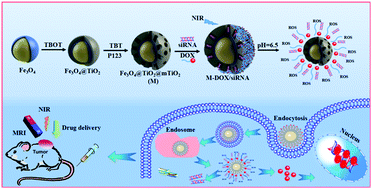
Photodynamic therapy (PDT), by producing reactive oxygen species (ROS), inhibits cancer cells and is an emerging and pioneering cancer therapeutic modality which can eliminate some of the drawbacks of other traditional anticancer therapies. To combine near-infrared (NIR) mediated PDT, chemotherapy and gene therapy in a synergistic manner, a novel NIR light activated photosensitizer for PDT was designed based on TiO2-coated Fe3O4 nanoparticle core/shell nanocarriers (Fe3O4@TiO2@mTiO2). The chemotherapeutic drug doxorubicin hydrochloride (DOX) was conjugated to the surface of the TiO2 mesopores through pH-reversible hydrazone bond linking and β-catenin siRNA was loaded in the mesopores. Fe3O4@TiO2@mTiO2–DOX/siRNA delivery systems have features and functions of magnetic targeting, fluorescence imaging, MRI diagnosis, and combination therapy through the simultaneous maneuvering of a magnet and NIR-mediated PDT. In vitro, Fe3O4@TiO2@mTiO2–DOX/siRNA effectively silences β-catenin gene, induces tumor cell apoptosis and consequently significantly enhances the cancer suppression effect of the synergistic therapeutic agent. Meanwhile, under NIR irradiation, excess ROS produced can further trigger tumor cell apoptosis. In vivo investigation confirmed that Fe3O4@TiO2@mTiO2–DOX/siRNA exhibited high tumor targeted specificity through MRI and fluorescence imaging, and optimal anti-tumor efficacy. The results verified its significant therapeutic effects on tumors by combination therapy consisting of magnetic targeting and NIR-mediated PDT.


 Please wait while we load your content...
Please wait while we load your content...