Tunable self-assembly of Irinotecan-fatty acid prodrugs with increased cytotoxicity to cancer cells†
Abstract
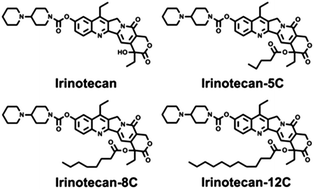
The development of a clinical chemotherapeutic is not an easy task. One challenge is how to deliver the agent to cancer cells. Nano-formulation of prodrugs, which combines the strengths of nanotechnology and prodrugs, possesses many advantages for chemotherapeutic drug delivery, including high drug loading efficiency, improved drug availability and enhanced accumulation in cancer cells. Here, we have constructed a small library of Irinotecan-derived prodrugs, in which the 20-hydroxyl group was derived with fatty-acid moieties through esterification. This conjugation fine-tuned the polarity of the Irinotecan molecule, thus enhancing the lipophilicity of the prodrugs and inducing their self-assembly into nanoparticles with different morphologies. These nano-formulated prodrugs accumulated at higher levels in cancer cells and were much more cytotoxic than free drugs. The rational design of prodrug-based nano-formulations opens a new avenue for the engineering of more efficient drug-delivery systems.
- This article is part of the themed collection: 2016 Journal of Materials Chemistry B Hot Papers

 Please wait while we load your content...
Please wait while we load your content...