Glucose oxidase-directed, instant synthesis of Mn-doped ZnS quantum dots in neutral media with retained enzymatic activity: mechanistic study and biosensing application†
Abstract
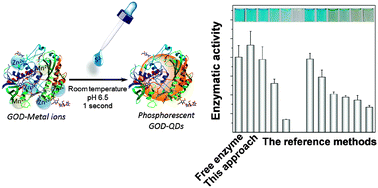
Protein-directed synthesis of quantum dots (QDs) is a “greener” alternative to the current high-temperature and aqueous synthetic protocols, which provide water-soluble, biocompatible protein-functionalized QDs in one-pot. However, the protein activity in such synthetic schemes is a critical issue, since the synthetic conditions (for instance, high pH of the precursors, long time of synthesis, and disruption of disulfide bonds) are not suitable for retaining the activity (especially for enzymes). Herein, we present a facile and instant glucose oxidase (GOD)-directed strategy for the preparation of highly luminescent, phosphorescent Mn-doped ZnS (Mn–ZnS) QDs in one-step at room temperature and in neutral aqueous media. With such mild synthetic conditions, the enzymatic activity of GOD was totally retained. Furthermore, we also carried out GOD-directed synthesis of QDs with several other conditions that are reported in the literature. It turned out that the GOD enzymatic activity under these synthetic conditions was lower than that of the proposed protocol, indicating that mild synthetic conditions are the prerequisite for retaining the enzymatic activity. Importantly, the as-prepared GOD-mediated Mn–ZnS QDs exhibited high photostability, high salt tolerance and colloidal stability, and can be stored for months at 4 °C or 25 °C without changing their phosphorescent intensity and enzymatic activity. Via selective chemical modification, the exact functional groups (amino acid residues) of GOD in directing the synthesis of Mn–ZnS QDs were studied in detail. It turned out to be imidazole in histidine residues but not thiol in cysteine residues that directed the formation of Mn–ZnS QDs, and this was further confirmed with several other proteins for synthesis of Mn–ZnS QDs. The as-prepared GOD-capped Mn–ZnS QDs were employed as phosphorescent probes for background-free sensing of glucose in serum samples.
- This article is part of the themed collection: 2015 Journal of Materials Chemistry B Hot Papers

 Please wait while we load your content...
Please wait while we load your content...