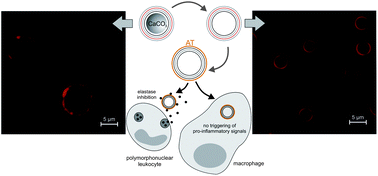
The treatment of chronic inflammation requires new concepts since recent approaches are mostly accompanied by massive side effects. Layer-by-layer (LbL) microcapsules, functionalized with anti-inflammatory substances such as α1-antitrypsin (AT), may avoid major side- and off-target effects thanks to their local application and the sustained delivery of defined amounts of the active agents into polymorphonuclear leukocytes (PMNs). However, LbL microcapsule application in inflamed tissues requires specific design and preparation. High biocompatibility has to be guaranteed by using biopolymers and ensuring the complete dissolution of the particle core. Moreover, off-target effects – such as macrophage-mediated pro-inflammatory signaling – have to be avoided. In our approach, biopolymer-coated CaCO3 particles were used and the core dissolution process was optimized to obtain highly biocompatible, non-aggregated, long-time stable and clearly calcium-free capsules. A fast verification tool was applied to monitor the remaining Ca2+ content. The incubation with macrophages shows reduced pro-inflammatory signaling compared to microparticles. Regarding their performance as a drug delivery system, AT-functionalized capsules showed a high inhibiting capacity towards neutrophil elastase, a major degradative enzyme in chronic inflammation. Consequently, the optimized design and preparation methods described in this study provide the basis for the development of medically applicable LbL carrier systems for active agents in the treatment of inflammatory processes.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...