Unveiling the role of carbon oxidation in irreversible degradation of atomically-dispersed FeN4 moieties for proton exchange membrane fuel cells†
Abstract
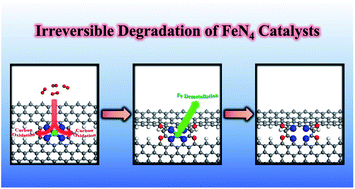
Nonprecious Fe–N–C catalysts containing atomically-dispersed FeN4 moieties are today the best candidates to replace platinum in proton exchange membrane fuel cell (PEMFC) cathodes. However, limited understanding of problematic operando degradation mechanisms in these catalysts largely impedes widespread commercialization. Recent experiments have shown that there exist durable and non-durable FeN4 sites in Fe–N–C catalysts for PEMFCs [J. Li et al., Nat. Catal., 2021, 4, 10–19]. Yet, the identification of which FeN4 sites are durable and which are not – and why – remains unclear. Using first-principles density functional theory (DFT) computations, we investigated the irreversible degradation of FeN4 catalysts at the atomic level, caused by Fe de-metalation and chemical oxidation of carbon via a proposed new carbon oxidation pathway. Our computational results show that oxidation of surface carbon next to FeN4 moieties at interior sites is essentially reversible under operando electrochemical conditions; whereas oxidation of carbon next to FeN4 moieties at the edge sites leads to accelerated Fe de-metalation, inducing irreversible degradation of FeN4 catalysts. From amongst six FeN4 moieties established experimentally, we identify three durable and three non-durable configurations. This work resolves the controversy as to which FeN4 moieties are durable under PEMFC operando conditions and provides a deeper understanding of the irreversible degradation mechanism of FeN4 catalysts in acidic media, furnishing a practical guide for rational design of FeN4 catalysts with long-term durability.


 Please wait while we load your content...
Please wait while we load your content...