A two-dimensional conductive Mo-based covalent organic framework as an efficient electrocatalyst for nitrogen fixation†
Abstract
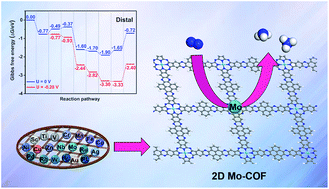
Electrocatalytic reduction of nitrogen (N2) is considered as a simple, green, and sustainable method for producing ammonia (NH3). Inspired by the recent experimental synthesis of a two-dimensional intrinsically conductive Ni-based covalent organic framework (Mirica et al., J. Am. Chem. Soc., 2019, 141, 11929–11937), here, on the basis of density functional theory (DFT), we explore the electrocatalytic performance of a series of stable and conductive two-dimensional (2D) TM-based covalent organic frameworks (TM-COFs, TM = Sc, Ti, V, Cr, Mn, Fe, Co, Ni, Cu, Zn, Nb, Mo, Ru, Rh, Pd, Ag, W, Ir, Pt, and Au, respectively) toward the N2 reduction reaction (NRR), which are constructed by the robust linkage between 2,3,9,10,16,17,23,24-octaamino-metallophthalocyanine and pyrene-4,5,9,10-tetraone. The computational results show that the 2D conductive Mo-COF exhibits the highest electrocatalytic performance for N2 fixation with a very low overpotential of 0.16 V among the 20 candidates, and can effectively inhibit the competitive hydrogen evolution reaction (HER). The outstanding NRR activity and selectivity of the Mo-COF stem from its inherent superiorities, such as excellent electrical conductivity, significant positive charge and large spin moment on the Mo atom, and appropriate adsorption strength for NRR species. This work will promote more experimental research in this field to discover more highly active COF-based catalysts for advancing sustainable NH3 production.


 Please wait while we load your content...
Please wait while we load your content...