What is the better choice for Pd cocatalysts for photocatalytic reduction of CO2 to renewable fuels: high-crystallinity or amorphous?†
Abstract
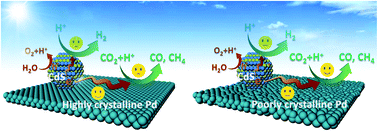
Solar-driven photocatalytic conversion of CO2 to renewable fuels represents an appealing approach to remedy environmental damage and address the energy crisis. Recently, the photocatalytic conversion efficiency has been improved with the implantation of cocatalysts into semiconductor photocatalysts. However, catalytic activity and selectivity are often limited by the uncontrollable charge trapping and surface reactivity behaviors of cocatalysts. Herein, for the first time, we reveal that engineering the crystallinity of cocatalysts offers an efficient route to tune both the interfacial charge kinetics and surface reaction dynamics. With Pd nanosheets as model cocatalysts, the influence of the degree of crystallinity of Pd on the photocatalytic performance is systematically investigated. When integrated with CdS quantum dots, it is found that high-crystallinity Pd with prefect lattice periodicity and fewer bulk and interfacial defects is conducive to the electron transfer from CdS to Pd, which thus yields more effective inhibition of H2 evolution on the CdS surface and achieves the highest selectivity of 100% for CO2 reduction. In contrast, low-crystallinity Pd provides large numbers of surface unsaturated atoms and defects as highly active sites for highly efficient CO2-to-CO/CH4 conversion. The combination of experimental analysis with theory simulation suggests that the amorphous Pd outperforms the high-crystallinity one in the adsorption and activation of CO2 molecules. As a result, the CdS–Pd sample with the largest percentage of amorphous domains of Pd achieves the highest CO and CH4 evolution rates, 10.3 and 5.9 times higher than those of pristine CdS, respectively. This work opens a new avenue to tune the photocatalytic activity and selectivity in solar driven CO2 conversion through regulating the crystallinity of cocatalysts.


 Please wait while we load your content...
Please wait while we load your content...