Unraveling nanoscale electrochemical dynamics of graphite fluoride by in situ electron microscopy: key difference between lithiation and sodiation†
Abstract
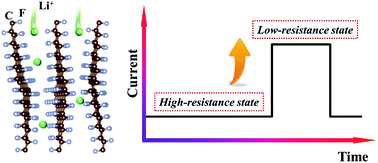
Graphite fluoride (CFx) is a particularly important cathode material for lithium primary batteries owing to its ultra-high theoretical capacity and energy density. However, a fundamental understanding of the electrochemical lithiation dynamics of CFx compounds remains sufficiently elusive at the nanoscale level. Here we present the use of an in situ transmission electron microscope (TEM) for studies of the nanoscale dynamics of individual CFx nanosheets upon electrochemical lithiation as well as sodiation. Importantly, while the sodiation reaction of CFx undergoes a conventional stepwise-propagation process, a mechanistically distinct two-stage process was unraveled for the lithiation reaction. This anomalous two-stage process consists of an initial slow lithiation stage where the CFx nanosheet is at a high-resistance state (HRS) and a subsequent ultra-fast lithiation stage with sharply increased current that corresponds to a low-resistance state (LRS) due to the formation of percolative conduction paths of pure carbon phases inside the CFx nanosheet. These mechanistic insights may provide a rational framework for the development and optimization of Li/CFx and Na/CFx batteries.


 Please wait while we load your content...
Please wait while we load your content...