Lithium-desorption mechanism in LiMn2O4, Li1.33Mn1.67O4, and Li1.6Mn1.6O4 according to precisely controlled acid treatment and density functional theory calculations†
Abstract
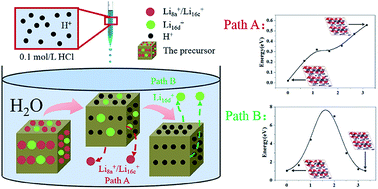
Spinel-type lithium manganese oxides (LMOs) are the most promising lithium-adsorption materials. LMOs can be in the form of solid solutions of LiMn2O4, Li1.33Mn1.67O4, and Li1.6Mn1.6O4. However, uncertainty about the lithium-desorption mechanism restricts material development. The synthesis of intermediate products of spinel-type LMOs during acid treatment is important to investigate such a mechanism. In this work, precisely controlled acid treatment experiments were performed to successfully obtain the intermediate products of LiMn2O4, Li1.33Mn1.67O4, and Li1.6Mn1.6O4. For LiMn2O4, lithium was desorbed with the dissolution of manganese. For Li1.33Mn1.67O4 and Li1.6Mn1.6O4, the lithium-desorption mechanism was an ion-exchange reaction, where lithium ions (Li+) in the 8a sites were prioritized for the ion-exchange reaction compared with Li+ in the 16d sites. Density functional theory (DFT) calculation results confirmed the experimental results and explained that the prioritization of Li+ in the 8a sites arose from its lower reaction-energy barrier, thereby revealing that Li+ preferred to move to the nearest vacant 8a sites when lithium was desorbed because of the lower diffusion-energy barrier. This work is the first to clarify the lithium-desorption mechanism through experiments and DFT calculations, and thus lays a foundation for the further exploitation of LMOs.


 Please wait while we load your content...
Please wait while we load your content...