Graphene–boron nitride hybrid-supported single Mo atom electrocatalysts for efficient nitrogen reduction reaction†
Abstract
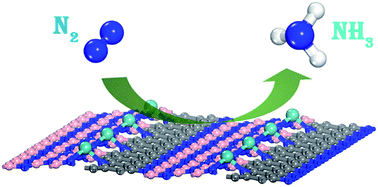
The conversion of nitrogen into ammonia under ambient conditions is of great significance but remains a great challenge. Interfacing of graphene with hexagonal boron-nitride (h-BN) leads to the formation of a hybrid sheet, namely BCN, which can be used as a stable and conductive support for an efficient single atom catalyst (SAC). Using first principles calculations, we designed a single Mo atom anchored on a defective BCN monolayer (Mo@BCN) as a durable, highly stable, and efficient SAC for the nitrogen reduction reaction (NRR). Compared to the semiconducting Mo-anchored h-BN, Mo@BCN exhibited metallic property that favored charge transport to active sites to drive the NRR. Importantly, this hybrid catalyst promoted the dominant NRR while suppressing the competitive hydrogen evolution reaction (HER). Investigation of the detailed mechanisms, including distal, alternating, and enzymatic mechanisms, for the reduction of N2 to NH3 has revealed outstanding catalytic performance of Mo@BCN at the quite low overpotential of 0.42 V via the enzymatic pathway independent of carbon concentration; thus, this study would lead to a new strategy for the development of efficient and stable SACs using BN-based materials as the substrate.


 Please wait while we load your content...
Please wait while we load your content...