Dual-nitrogen-source engineered Fe–Nx moieties as a booster for oxygen electroreduction†
Abstract
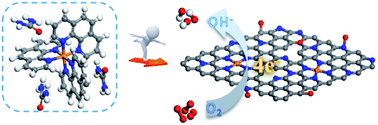
Metal–air batteries, particularly Zn–air batteries, have triggered considerable enthusiasm of communities due to their high theoretical power density. Developing highly active, cost-effective and alternative non-precious metal catalysts for the oxygen reduction reaction (ORR) is pivotal for popularizing zinc–air batteries. The rational design and synthesis of this type of catalyst are therefore critical, but it is still challenging to control the well-defined active sites as expected. Herein, we report a dual-nitrogen-source mediated route for synergistically controlling the formation of active Fe–Nx moieties that are embedded in the carbon matrix. The facile control of coordination structures of precursors by this dual-nitrogen-source approach is revealed to play a key role in this report. Impressively, the optimized dual-nitrogen-source derived catalyst (i.e. Fe–N–C-800) exhibits prominently enhanced ORR activity with a half-wave potential of 0.883 V in alkaline electrolyte, higher by 32 mV and 72 mV than those derived from individual nitrogen sources, which is also further evaluated in primary Zn–air batteries. The enhanced ORR activity of Fe–N–C-800 is attributed to the rich Fe–Nx active sites derived from the dual-nitrogen-source approach.


 Please wait while we load your content...
Please wait while we load your content...
