Porous oxygen vacancy-rich V2O5 nanosheets as superior semiconducting supports of nonprecious metal nanoparticles for efficient on-demand H2 evolution from ammonia borane under visible light irradiation†
Abstract
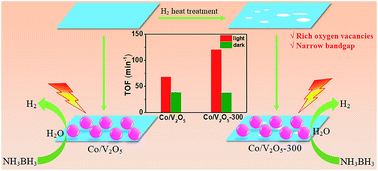
From the perspective of controlling visible-light-driven catalytic activities of supported metal catalysts by tailoring the band structures of semiconducting supports, we calcined pristine V2O5 in a H2 atmosphere at 250–350 °C to synthesize a series of porous two-dimensional V2O5 nanosheets with narrow bandgaps and rich oxygen vacancies, which supported Co and Ni nanoparticles as catalysts for visible-light-driven on-demand H2 evolution from ammonia borane (NH3BH3) in aqueous solution at 25 °C. The systematic investigations showed that all the catalysts had higher activities under visible light irradiation than in the dark and one Co catalyst exhibited the highest photocatalytic activity with a total turnover frequency (TOF) value of 120.4 min−1, which exceeded the values of all the reported noble metal-free catalysts. Structural characterization indicated that pristine V2O5 and the three post-modified V2O5 species had different bandgaps, valence band positions and concentrations of oxygen vacancies as well as different photocurrent densities, leading to different electron densities of active metal NPs in the supported catalysts with different catalytic activities. This was verified using the UV-vis spectra, valence band X-ray photoelectron spectroscopy, X-ray diffraction patterns and electrochemical characterization of various V2O5 species and the corresponding photocatalytic performance of catalysts.


 Please wait while we load your content...
Please wait while we load your content...