Facile synthesis of Co/Pd supported by few-walled carbon nanotubes as an efficient bidirectional catalyst for improving the low temperature hydrogen storage properties of magnesium hydride†
Abstract
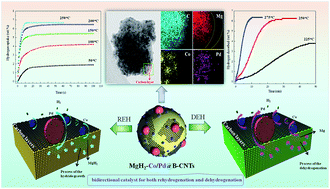
Catalytic doping is important for enhancing the hydrogen storage performance of metal hydrides, but it is challenging to develop a single catalyst to enhance both hydrogen desorption and absorption to a certain degree. Herein, a bidirectional Co/Pd catalyst, homogeneously loaded on bamboo-shaped carbon nanotubes (Co/Pd@B-CNTs), showed superior catalytic effects, improving both the hydrogen desorption and absorption properties of MgH2 at relatively low temperatures. The MgH2–Co/Pd@B-CNTs composite starts to release hydrogen at 198.9 °C, which is 132.4 °C lower than as-milled MgH2. The hydrogen desorption activation energy for MgH2 is reduced from 178.00 to 76.66 kJ mol−1 by the catalytic effects of Co/Pd@B-CNTs. The MgH2–Co/Pd@B-CNTs composite shows dramatically improved absorption kinetics; it rapidly uptakes 6.68 wt% H2 within 10 s at 250 °C, and quickly absorbs 1.91 wt% H2 within 100 s, even at a temperature as low as 50 °C. More importantly, a special mechanism for the “bidirectional catalyst” Co/Pd is proposed for the first time and discussed in detail. During the hydrogenation process, elemental Pd plays a dominant role in accelerating the preferential diffusion of hydrogen atoms at the Pd/Mg interface, while during dehydrogenation, phase transformation between Mg2Co and Mg2CoH5 as well as a Mg–Pd alloy becomes the crucial factor, facilitating the release of hydrogen atoms by decreasing the diffusion barrier. Moreover, novel structures of bamboo-shaped carbon nanotubes with a large diameter (>100 nm) and high specific surface area (146.8 m2 g−1) allow the homogenous dispersion of Co/Pd NPs and enhance the direct contact with MgH2 particles.
- This article is part of the themed collection: 2019 Journal of Materials Chemistry A HOT Papers


 Please wait while we load your content...
Please wait while we load your content...