Polyoxometalate-assisted formation of CoSe/MoSe2 heterostructures with enhanced oxygen evolution activity†
Abstract
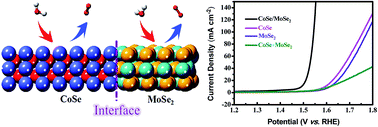
The oxygen evolution reaction (OER) is a half reaction of electrochemical water splitting that suffers from a kinetically sluggish four-electron process, and it is regarded as the efficiency-limiting step in water splitting. Herein, heterostructures of CoSe (cobalt selenide) nanoparticles and MoSe2 (molybdenum selenide) nanosheets (CoSe/MoSe2 hybrids) were fabricated through a non-metal-induced growth method. Due to the increase in the effective specific area and the electron transfer ability caused by the formation of the heterogeneous interface, the obtained CoSe/MoSe2 hybrids show superior OER performance (η = 262 mV at 10 mA cm−2) and long-term stability (20 h for continuous testing) as compared to pure CoSe, MoSe2 and physically mixed CoSe and MoSe2. Schematic energy band diagrams derived from ultraviolet photoelectron spectroscopy results further confirmed the electronic modulation between CoSe and MoSe2 and revealed that the d-band center of CoSe/MoSe2 hybrids moved closer to the Fermi level, giving rise to high charge carrier density and low intermediate adsorption energy as compared to CoSe and MoSe2. This work provides some insight into the design and synthesis of heterostructured nanomaterials from the MOF precursors.


 Please wait while we load your content...
Please wait while we load your content...