Designing oxygen bonding between reduced graphene oxide and multishelled Mn3O4 hollow spheres for enhanced performance of supercapacitors†
Abstract
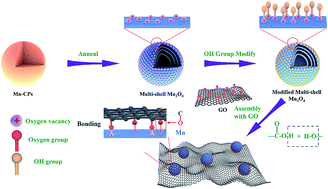
Strong chemical bonds between transition metal oxides and carbon materials which enable fast electron transfer kinetics are highly required in supercapacitor electrodes. Meanwhile, hollow transition metal oxide nanostructures have promising potential for advanced energy storage applications. Herein, an oxygen vacancy assisted hydroxyl modification method is proposed to encapsulate Mn3O4 hollow spheres with controlled shell numbers in reduced graphene oxide (Mn3O4–rGO). The strong bonds between Mn3O4 and graphene result in fast charge transport, uniform distribution, increased active sites and enhanced structural stability. Originating from the structural features, when these materials were evaluated as electrodes for supercapacitors, Mn3O4–rGO shows a high specific capacitance of 561.5 C g−1 with good rate performance and excellent stability with 98% retention after 10 000 cycles. In order to widen the potential application of supercapacitors, asymmetric supercapacitors (ASCs) are assembled using Mn3O4–rGO as positive electrodes and active carbon as negative electrodes. The ASCs exhibit a high specific capacitance of 180.2 F g−1, delivering a high energy density of 72.3 W h kg−1 at a power density of 864 W kg−1. This surface-confined strategy may pave a way for realizing strong chemical bonds between hollow oxides and carbon-based materials for high performance supercapacitors.


 Please wait while we load your content...
Please wait while we load your content...