Pb(ii), Cu(ii) and Cd(ii) removal using a humic substance-based double network hydrogel in individual and multicomponent systems†
Abstract
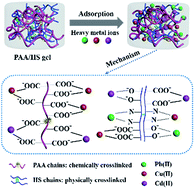
A systematic study of the interaction between adsorbents and potentially toxic metal ions in multicomponent systems is of great significance due to the coexistence of multiple potentially toxic metal ions in the real water environment. In this study, a new humic substance-based double network hydrogel (PAA/HS gel) was produced by a radical polymerization method and applied to Pb(II), Cu(II) and Cd(II) removal in both individual and multicomponent systems under various conditions. The macroporous network structure (aperture ≈ 10 μm) and high water permeability of the PAA/HS gel could fully facilitate the active site exposure and increase the potentially toxic metal ion diffusion rate. The PAA/HS gel exhibited high theoretical adsorption capacities of 360.50, 151.00 and 412.76 mg g−1 for Pb(II), Cu(II) and Cd(II) in individual systems. Noticeably, both interference and promotion of adsorption behaviors were identified in multicomponent systems. In binary systems, the adsorption capacity and rate were decreased for Pb(II) and Cu(II), while increased for Cd(II). In ternary systems, the adsorption capacity and rate for Pb(II), Cu(II) and Cd(II) were decreased. The adsorption isotherms and kinetics were well fitted by Langmuir–Freundlich and Elovich models, respectively, indicating that the PAA/HS gel possessed a heterogeneous surface and various types of binding sites. The FTIR and XPS studies confirmed that both nitrogen- and oxygen-containing functional groups (i.e., amino, phenolic hydroxyl and carboxyl groups) were involved in the adsorption process, while the interactions between functional groups and Pb(II), Cu(II) and Cd(II) were different.
- This article is part of the themed collection: Materials and Nano Research in Atlanta


 Please wait while we load your content...
Please wait while we load your content...