Chemical reactions under the nanofluidic confinement of reconstructed lamellar membranes†
Abstract
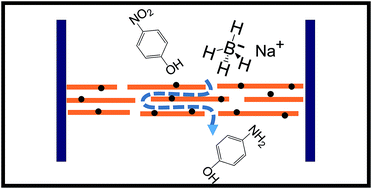
The nanochannel network of reconstructed vermiculite clay is employed here to study the effects of nanofluidic confinement on chemical reactions. The nanofluidic confinement not only improved the efficiency of the palladium and gold nanoparticle assisted catalytic reduction of nitro-arene compounds, but also reduced the concentration of sodium borohydride required for the reactions. The augmentation of reaction rate inside the nanofluidic channels is attributed to the trapping of volatile hydride ions, enhanced local concentrations of the reactants and continuous elimination of the product from the nanochannel reactor. The effects of nanofluidic confinement were found to increase with increasing channel lengths and decreasing channel heights. The ease of preparation and modification of reconstructed layered material based membranes makes them an ideal candidate for studying the true effects of nanofluidic confinement.


 Please wait while we load your content...
Please wait while we load your content...