Open hollow Co–Pt clusters embedded in carbon nanoflake arrays for highly efficient alkaline water splitting†
Abstract
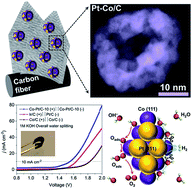
Water splitting provides a clean and renewable way to produce high-purity hydrogen; the current bottlenecks of which are the slow kinetics and poor stability of the available electrocatalysts. Herein, we report a class of open hollow Co–Pt bimetallic nanoclusters embedded in mesoporous N-doped carbon nanoflake arrays grown on flexible carbon cloth (Co–Pt/C NAs, 2.5 wt% Pt), which demonstrate outstanding electrocatalytic activity and ultra-long durability for both hydrogen and oxygen evolution in alkaline media (50 mV for hydrogen evolution and 320 mV for oxygen evolution, vs. RHE). The high performance arises from the unique nanostructure and the synergy of Co–Pt promoting water dissociation as demonstrated by density functional theory (DFT) calculation results. It can be directly utilized as a highly efficient bifunctional electrocatalyst for overall alkaline water splitting, and it outperforms noble-metal-based materials in terms of much lower operating voltage (1.54 V at 10 mA cm−2) and higher stability (no degradation at a constant current or voltage up to 120 h), representing a highly promising electrode for electrochemical energy conversion.


 Please wait while we load your content...
Please wait while we load your content...