Tailoring the nano heterointerface of hematite/magnetite on hierarchical nitrogen-doped carbon nanocages for superb oxygen reduction†
Abstract
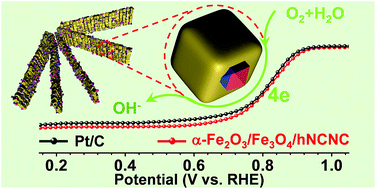
Nowadays the main challenge for large-scale applications of fuel cells is the designing of cheap and stable electrocatalysts for the oxygen reduction reaction (ORR). Herein, we report a convenient approach to a highly active cheap ORR electrocatalyst by tailoring the nano heterointerface of α-Fe2O3/Fe3O4 on hierarchical nitrogen-doped carbon nanocages (hNCNC) via the partial carbothermal reduction of α-Fe2O3 nanocrystals to Fe3O4. The so-constructed heterostructural α-Fe2O3/Fe3O4/hNCNC electrocatalyst exhibits excellent ORR performance surpassing commercial Pt/C, with a high onset potential (1.03 V vs. reversible hydrogen electrode) and half-wave potential (0.838 V), large limiting current density (6.02 mA cm−2 at 0.5 V), and robust durability and methanol tolerance. The heterostructure is identified by high-angle annular dark field scanning transmission electron microscopy coupled with electron energy-loss spectroscopy. Acid leaching experiments and theoretical calculations indicate that the heterostructural α-Fe2O3/Fe3O4 species with variable Fe valences are responsible for the high ORR performance. In principle, regulating the nano heterointerface based on the multivalent metal species in a nanocrystal matrix could become a new convenient approach to developing high-performance electrocatalysts or even various functional materials.


 Please wait while we load your content...
Please wait while we load your content...