Oxygen vacancy-originated highly active electrocatalysts for the oxygen evolution reaction†
Abstract
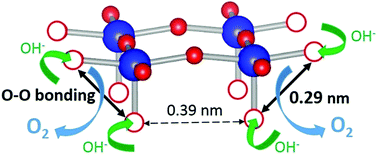
The oxygen evolution reaction (OER) is an important reaction in the field of renewable energy and is utilized in electrochemical water splitting for hydrogen fuel production and rechargeable metal–air batteries. Herein, we report a new oxygen evolution reaction mechanism originating from oxygen vacancies, which remarkably enhances the OER activity of oxygen deficient electrocatalysts. The OER activity of Sr2VFeAsO3−δ is drastically enhanced above the lattice oxygen vacancy of δ = 0.5, exhibiting ∼300 mV lower overpotential and 80 times higher specific activity at 1.7 V vs. RHE. Surprisingly, the initially low OER activity of Sr2VFeAsO3−δ (δ < 0.5) is enhanced to the level of state-of-the-art OER catalysts simply by introducing higher concentrations of oxygen vacancies. Density functional theory (DFT) calculations clarify that the oxygen vacancies are initially dominated by one of the crystallographic sites, while two types of crystallographic sites become fully accessible for δ > 0.5. As a result, the distance between OH− coupled oxygen-vacant sites becomes sufficiently short to enable direct O–O bond formation as in photosystem II. Thus, we found that the OER activity of oxygen deficient electrocatalysts is controllable by the variety of lattice oxygen vacancies, which suggests that oxygen deficient layered superconductors are promising OER catalysts for energy conversion technologies.
- This article is part of the themed collection: Industry R&D collection


 Please wait while we load your content...
Please wait while we load your content...