Synergetic co-immobilization of SeO42− and Sr2+ from aqueous solution onto multifunctional graphene oxide and carbon-dot based layered double hydroxide nanocomposites and their mechanistic investigation†
Abstract
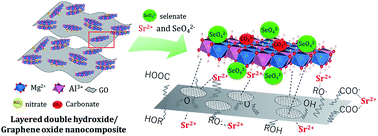
The co-immobilization of radioactive Sr2+ and SeO42− using a multifunctional material is an interesting area of research for the total remediation of radioactive wastes. However, it is rather challenging to target both the anion and cation through the realization of a multifunctional ability of the resultant sorbent. In this work, MgAl–NO3-layered double hydroxides (LDHs) containing graphene oxide (GO) and carbon-dot (C-dot) nanocomposites were synthesized and contrasted for the co-immobilization of Sr2+ and SeO42−. Zeta potential measurements and TEM observation of the MgAl–NO3-LDH/C-dot composite indicated that the carbon-nanodot was attached to the surface of LDH nanosheets, while in the MgAl–NO3-LDH/GO composites the LDH nanosheets were decorated on the larger sized GO nanosheets. Adsorption studies found that the normalized Sr2+ adsorption capacity was 1.793 mmol g−1 on MgAl–NO3-LDH/GO, which was nine times higher than that for the MgAl–NO3-LDH/C-dot composite and GO. The enhancement in the Sr2+ adsorption capacity is due to the co-operative effect of the LDH and GO. The adsorption of Sr2+ on the MgAl–NO3-LDH/C-dot occurs by co-ordination with the –COO− group, while ligand exchange and ionic interaction with the alkoxide anion are the dominant mechanisms on the MgAl–NO3-LDH/GO composite. Moreover, the adsorption capacity of both Sr2+ and SeO42− increased synergistically in the bi-component system containing both ions. The present technique is promising and offers a sustainable and environmentally friendly multifunctional material for the co-immobilization of both anionic and cationic radioactive surrogates from aqueous solutions.


 Please wait while we load your content...
Please wait while we load your content...