The origin of excellent rate and cycle performance of Sn4P3 binary electrodes for sodium-ion batteries†
Abstract
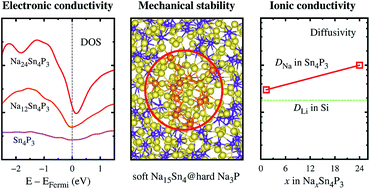
Despite numerous studies on binary electrodes for Na-ion batteries, a fundamental understanding of their superior performance is lacking. We clarify the sodiation process of a promising Sn4P3 anode and the origin of its outstanding performance. The Na ions preferentially react with P rather than Sn. Key products formed in NaxSn4P3 during sodiation are local Sn particles around x = 4, a Na3P subphase above x = 8, and a Na15Sn4 subphase above x = 12. The preference of Na for P and the stable formation of identified products are driving forces for the formation of a characteristic morphology where Na15Sn4 nanoparticles are embedded in a Na3P matrix. This morphology has high structural integrity because Na3P has a 71% higher bulk modulus than Na15Sn4 and thus the hard Na3P matrix can effectively protect the soft Na15Sn4 cores. Among the incoming electrons into Sn4P3 during sodiation, 76% and 24% are stored in Sn and P, respectively, and those stored in Sn boost the electron density at the Fermi level, significantly improving the electronic conductivity. The Na ions in Sn4P3 diffuse very rapidly regardless of the Na concentration, and they diffuse faster than the Li ions in Si. The fast Na ion conductivity results from the facilitation of the bond breaking/making process due to the richness of weak Na–Sn bonds surrounding a diffusing Na ion. This study offers guidelines for designing superior binary anode materials for Na-ion batteries.


 Please wait while we load your content...
Please wait while we load your content...