Oxygen-deficient WO3−x@TiO2−x core–shell nanosheets for efficient photoelectrochemical oxidation of neutral water solutions†
Abstract
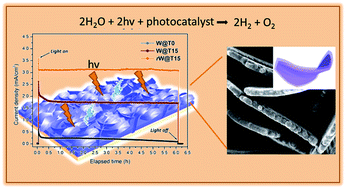
Preparation of highly active, stable and earth-abundant photoanodes for water oxidation is an important strategy to meet the demand of developing clean-energy technologies. In this paper, efficient and stable photoanodes based on oxygen-deficient black WO3−x@TiO2−x core–shell nanosheets with precisely controlled shell thickness have been fabricated for photoelectrochemical (PEC) conversion from neutral water solutions. The black WO3−x@TiO2−x core–shell nanosheet photoanode with the shell thickness of ∼15 nm achieved around 8 times higher photocurrent density (∼3.20 mA cm−2) than the pure WO3 photoanode at 1.23 V vs. the RHE. An improved onset potential with long-term PEC durability was also realized with the obtained black WO3−x@TiO2−x core–shell nanosheet photoanodes. The promoted PEC water oxidation performance was likely to be originated from enhanced light absorption, interfacial charge transfer and charge separation in these WO3−x@TiO2−x nanosheets which were revealed by finite-difference time-domain simulations and specific band alignment, along with optical and electrochemical spectroscopic evidence. In a word, such black WO3−x@TiO2−x nanosheet photoanodes suggest many exciting opportunities for PEC water splitting toward highly efficient solar fuel generation and many other PEC sensing applications.


 Please wait while we load your content...
Please wait while we load your content...