Bis(tri-n-alkylsilyl oxide) silicon phthalocyanines: a start to establishing a structure property relationship as both ternary additives and non-fullerene electron acceptors in bulk heterojunction organic photovoltaic devices†
Abstract
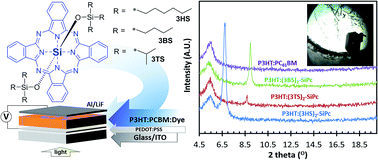
Previous studies have shown that the use of bis(tri-n-hexylsilyl oxide) silicon phthalocyanine ((3HS)2-SiPc) as a solid ternary electroactive additive in poly(3-hexylthiophene):phenyl-C61-butyric acid methyl ester P3HT:PC61BM bulk heterojunction organic photovoltaic (BHJ OPV) devices resulted in an increased performance. It has been hypothesized that the increase in efficiency is partially due to the unique and odd combination of high solubility and strong driving force of crystallization previously observed for (3HS)2-SiPc. In this follow-up study, two chemical variants of (3HS)2-SiPc, namely bis(tri-n-butylsilyl oxide) ((3BS)2-SiPc) and bis(tri-n-isopropylsilyl oxide) ((3TS)2-SiPc) were synthesized to determine how small changes in the chemical structure would affect the properties of the material and its performance within BHJ OPV devices. We observed that the use of either (3XS)2-SiPc compound results in a further ∼10% increase in JSC compared to the use of (3HS)2-SiPc. We also did a preliminary assessment of the use of three (3XS)2-SiPcs as replacements for PC61BM in straight binary P3HT-based BHJ OPV devices. Despite achieving only ∼1% PCE efficiencies, observations including a ≈50% increase in VOC over a P3HT:PC61BM baseline and a decent fill factor indicate to us that (3XS)2-SiPcs do have potential as non-fullerene acceptors and advantageous alternatives due to their low embedded energy and therefore their inherent sustainability. X-ray diffraction of ternary and binary BHJ devices demonstrates that both (3BS)2-SiPc and (3TS)2-SiPc experienced similar increase in crystallite density (d-spacing) relative to (3HS)2-SiPc which we surmise plays a role in the improved device efficiency. Like (3HS)2-SiPc, for these two new additives, we also observed a high tendency of crystallization. The results from this study suggest that solubility and driving force to crystallize are important factors in determining the extent to which an additive will migrate to the donor/acceptor interface and thus affect its performance as a ternary additive in BHJ OPV devices. Based on the three (3XS)2-SiPcs used in this study, the smaller tri-n-alkylsilyl oxide molecular fragments seem to work better. Therefore, moving forward, we will continue to consider smaller molecular fragments that still enable solubility and processability of (3XS)2-SiPcs.


 Please wait while we load your content...
Please wait while we load your content...