Synergistic effects towards H2 oxidation on the Cu–CeO2 electrode: a combination study with DFT calculations and experiments†
Abstract
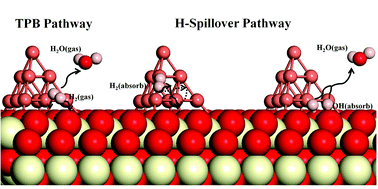
The solid oxide fuel cell (SOFC) is an environmentally-friendly, highly efficient, and fuel adaptable electrochemical conversion device. The Cu–CeO2 material has been recognized as a promising anode material for SOFCs. Although Cu is not a good enough oxidation catalyst, and has a relatively low catalytic activity, a combination of Cu and CeO2 can strengthen catalytic activity and overcome the problems associated with either Cu or CeO2 individually. Our density functional theory (DFT) calculations illustrate that a Cu cluster supported on CeO2(111) suppresses the formation of interface oxygen vacancies, while also enhancing the catalytic activity, and reduces the energy barrier of the H2 oxidation reaction process compared to that of stoichiometric CeO2(111). The three phase boundary (TPB) pathway with the highest energy barrier of 0.836 eV is obviously much lower than the stoichiometric CeO2(111) equivalent with the highest energy barrier of 2.399 eV. Experimentally, temperature programmed reduction (TPR) experiments establish that Cu particles can reduce the reduction reaction temperature for ceria and increase the amount of reduced ceria. In addition, a dramatic comparison between pure ceria and Cu-modified ceria through electrical conductivity relaxation (ECR) experiments quantitatively demonstrates that Cu particles greatly improve the reaction kinetics with the specific oxygen surface exchange coefficient increasing from 1.012 × 10−4 cm s−1 of the bare ceria to 12.180 × 10−4 cm s−1 of the Cu-modified ceria (CeO2–Cu80), which agrees well with the results of the theoretical calculations.

 Please wait while we load your content...
Please wait while we load your content...