Iron–cobalt bimetal decorated carbon nanotubes as cost-effective cathode catalysts for Li–O2 batteries†
Abstract
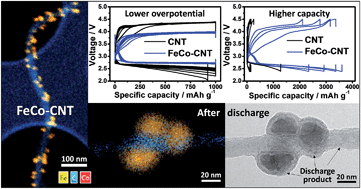
Despite the extremely high theoretical specific capacity of lithium oxygen (Li–O2) electrochemistry, low energy efficiency resulting from the large potential gap between the discharge and charge makes this system impractical. In this report, an iron cobalt bimetal decorated carbon nanotube (FeCo–CNT) composite was synthesized as a catalytic air cathode material for Li–O2 batteries. An Li–O2 battery using FeCo–CNT air electrodes exhibited higher efficiency (72.15%) than that of pristine CNTs (62.57%) as well as higher capacity (3600 mA h g−1vs. 1276 mA h g−1). Spectroscopic and electron microscopy analyses showed that the improved cell performances can be attributed to the catalytic effect of FeCo. As cost-effective non-noble metal catalysts, FeCo–CNTs demonstrated performance comparable to noble metal catalysts in Li–O2 systems.
- This article is part of the themed collection: Emerging Investigators 2016: Novel design strategies for new functional materials

 Please wait while we load your content...
Please wait while we load your content...