Biomass-derived activated carbon with simultaneously enhanced CO2 uptake for both pre and post combustion capture applications†
Abstract
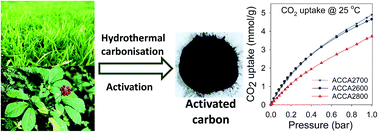
We report on the synthesis and CO2 uptake capabilities of a series of activated carbons derived from the biomass raw materials Jujun grass and Camellia japonica. The carbons were prepared via hydrothermal carbonization of the raw materials, which yielded hydrochars that were activated with KOH at temperature between 600 and 800 °C. Carbons activated at KOH/hydrochar ratio of 2 have moderate to high surface area (1050–2750 m2 g−1), are highly microporous (95% of surface area arises from micropores, and 84% of pore volume from micropores of size between 5 and 7 Å), and exhibit excellent CO2 uptake capacity at 25 °C of up to 1.5 mmol g−1 at 0.15 bar and 5.0 mmol g−1 at 1 bar, which is amongst the highest reported so far for biomass-derived carbons. On the other hand, activation at KOH/hydrochar ratio of 4 generates carbons with surface area and pore volume of up to 3537 m2 g−1 and 1.85 cm3 g−1, and which, depending on level of activation, simultaneously exhibit high CO2 uptake at both 1 bar (4.1 mmol g−1) and 20 bar (21.1 mmol g−1), i.e. under conditions that mimic, respectively, post combustion and pre combustion CO2 capture from flue gas streams. The present carbons are the first examples of biomass derived porous materials with such all-round CO2 uptake performance, which arises due to the pore size distribution of the carbons being shifted towards small micropores even for samples with very high surface area. Thus the carbons satisfy the requirements for both low pressure (presence of small micropores) and high pressure (high surface area) CO2 uptake.

 Please wait while we load your content...
Please wait while we load your content...