Experimental and theoretical investigations of nitro-group doped porous carbon as a high performance lithium-ion battery anode†
Abstract
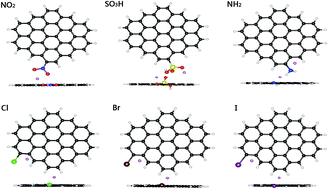
Doping is an effective solution to improve the capacity of carbon based anode materials such as introducing nitrogen, boron, sulfur, and phosphorus heteroatoms into the graphite lattice. However, most of the previous doping methods are confined to the crystal lattice and edge doping is rarely studied. Here, using first-principles quantum chemical calculations, we studied the lithium adsorption ability of various functional groups (NH2, NO2, SO3H, Cl, Br, I, OH, and P) which were doped at the edge of graphene sheets. Among all the groups, the nitro-group shows the best lithium adsorption properties. On the basis of theoretical predictions, we successfully synthesized nitro group edge modified porous carbon through the pyrolysis of Cu-based metal–organic frameworks (MOFs) at 600 °C under a nitrogen atmosphere and post-acid treatment. As an anode material for lithium ion batteries, it retains a capacity of 588 mA g−1 after 1500 cycles at a high current density of 1 A g−1. The lithium anodic performance of nitro-group doped carbon is superior to other edge doped carbon based materials reported in the literature such as halogen, sulfur and phosphorus. The excellent cycling performance at high current densities is ascribed to the improved lithium adsorption ability of the nitro-group doped at the edge of carbon.

 Please wait while we load your content...
Please wait while we load your content...