Full-molar-ratio synthesis and enhanced lithium storage properties of CoxFe1−xCO3 composites with an integrated lattice structure and an atomic-scale synergistic effect†
Abstract
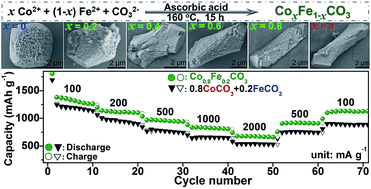
Multicomponent composites with an integrated lattice structure can possess an atomic-scale distribution of different components within the crystallites and may express an enhanced synergistic effect compared to the mechanical mixture. In this paper, a series of cobaltous-ferrous carbonates CoxFe1−xCO3 (x = 0, 0.2, 0.4, 0.6, 0.8 and 1) have been synthesized via a facile hydrothermal route with the assistance of ascorbic acid (AA). The four multicomponent CoxFe1−xCO3 composites exhibit hexagonal structures similar to those of the monocomponents FeCO3 and CoCO3, and the resulting rodlike crystallites give aspect ratios and unit-cell parameters linearly changed along with the increasing x value, indicating the successful synthesis of CoxFe1−xCO3 composites at an arbitrary Co/Fe molar ratio. When used as lithium ion battery anodes, the CoxFe1−xCO3 composites can well inherit both the high-conductivity characteristic of CoCO3 and the low-rate cycling stability of FeCO3. In particular, each composite presents better electrochemical properties than the corresponding xCoCO3 + (1 − x)FeCO3 mixture, mainly assigned to an inner atomic-scale synergistic effect within the formula CoxFe1−xCO3. Therefore, CoxFe1−xCO3 composites may serve as novel high-capacity LIB anode materials for practical application, and also a facile strategic approach is introduced for the full-molar-ratio synthesis of multicomponent composites.

 Please wait while we load your content...
Please wait while we load your content...