Ethanol-directed morphological evolution of hierarchical CeOx architectures as advanced electrochemical capacitors†
Abstract
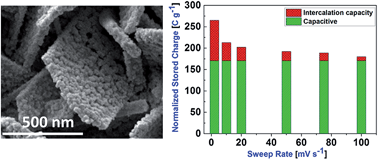
To surmount the fundamental limits of energy density in the current supercapacitor devices, the electrode materials should be capable of storing localized charge while having a high degree of freedom to provide ease of electron/ion flow in/out of the electrodes. Herein, we demonstrated a facile approach to designing CeOx based hierarchical architectures for high-performance energy storage electrodes. The unique CeOx based hierarchical architectures, including nanowires, nanocables, nano-micro biscuits and micro walls, were fabricated by simply manipulating the nitrate ion oxidation rate during electrochemical synthesis. Among all the electrodes, the CeOx nano-micro biscuits demonstrated the most excellent electrochemical performance from undergoing fast faradaic reactions leading to high specific capacitance within short charging time. Furthermore, voltammetric sweep mediated analysis was utilized to quantify the capacitive and intercalation effects in the total stored charge capacity of CeOx nano-micro biscuits. The presence of higher Ce3+ content (as confirmed by XPS studies) in CeOx nano-micro biscuits could be responsible for accelerating faradaic redox reactions (conversion of Ce3+ to Ce4+) in electrolyte solution, which is sufficient for realizing their superior performance over the other nano-microstructures. The present study demonstrates the effectiveness of CeOx as a low-cost and promising candidate for future energy storage/harvesting and integrated nanoelectronic devices.

 Please wait while we load your content...
Please wait while we load your content...