Magnéli phase Ti8O15 nanowires as conductive carbon-free energy materials to enhance the electrochemical activity of palladium nanoparticles for direct ethanol oxidation†
Abstract
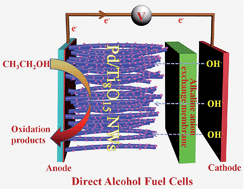
Pure single-crystalline Magnéli phase Ti8O15 nanowires (NWs) have been successfully synthesized via a facile one-step evaporation–deposition synthesis method in a hydrogen atmosphere. The electrical conductivity of a single Ti8O15 nanowire is 2060 S m−1 at 300 K, which is much higher than that of carbon black (∼100 S m−1) and almost as high as that of graphite (∼1000 S m−1) or graphene (∼2000 S m−1). Pd nanoparticles (NPs) loaded on Ti8O15 nanowires are synthesized through a pulsed electrodeposition method. As carbon-free support materials for Pd NPs, the Pd/Ti8O15 NWs show a significantly enhanced activity for the ethanol oxidation reaction and excellent durability compared to the Pd/C catalyst. The peak current density of the Pd/Ti8O15 NWs after 2000 cycles is 2791 mA mgPd−1, with only 18.6% loss, while, on the Pd/C electrode, only a current density of 682 mA mgPd−1 is observed, with a loss of 42.3% of the initial ethanol oxidation reaction activity. The synthetic approach for pure single-crystalline Magnéli phase Ti8O15 nanowires gives the inspiration for the development of conductive carbon-free materials for electrocatalysis in direct alcohol fuel cells (DAFCs).

 Please wait while we load your content...
Please wait while we load your content...