Preparation of titania based biocatalytic nanoparticles and membranes for CO2 conversion†
Abstract
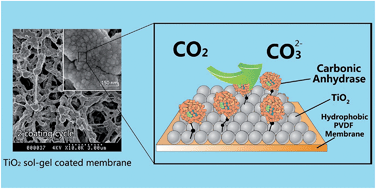
A biomimetic route for CO2 conversion catalyzed by carbonic anhydrase (CA) is an attractive option for carbon capture and storage due to the high efficiency and specificity of CA in CO2 hydration. The preparation of TiO2 based biocatalytic nanoparticles and membranes via CA immobilization facilitates the reuse of the enzyme and could be potentially integrated in a gas–liquid membrane contactor for highly efficient CO2 capture. In this work, different immobilization protocols were compared based on CA loading, activity and stability. For biocatalytic nanoparticles, over 80% activity recovery corresponding to 163 mg g−1 support was achieved. Repeated reuse and recovery of the biocatalytic nanoparticles over twenty cycles showed only modest loss in activity. For the biocatalytic membranes, the nanostructure of the titania coating and the pH values during immobilization were examined to optimize the biocatalytic performance. Biocatalytic membranes prepared at pH 6 with two cycles of sol–gel coating were able to immobilize a 700 μg CA per cm2 nominal membrane area. The CO2 hydration efficiency of the biocatalytic nanoparticles and membranes was examined, and only marginal loss of catalytic efficiency was observed when compared with their free CA counterpart, indicating good potential for application of such biocatalytic nanoparticles and membranes for CO2 conversion.
- This article is part of the themed collection: 2015 Journal of Materials Chemistry A Hot Papers

 Please wait while we load your content...
Please wait while we load your content...