Multiphase sodium titanate/titania composite nanostructures as Pt-based catalyst supports for methanol oxidation†
Abstract
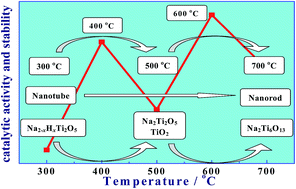
Sodium titanate/titania composite nanotubes/nanorods (STNS) are synthesized from anatase titania by the hydrothermal method and subsequent annealing in the range of 300–700 °C. The changes in the composition and morphology of STNS are investigated by X-ray diffraction (XRD) and transmission electron microscopy (TEM). The results reveal that the composition of STNS changes from “Na2−xHxTi2O5” to “Na2Ti6O13” and their morphology changes from nanotubes to nanorods. The products obtained at 400 °C and 600 °C correspond to the intermediate state of reactions. Pt-based catalysts are prepared by a microwave-assisted ethylene glycol process, and are also characterized by physical analysis and electrochemical measurements. The variations of the catalytic activity and stability of Pt/C-STNS catalysts show the interesting “M” shape with the increase of the annealing temperature of STNS. The Pt nanoparticles supported on STNS-400 nanotubes and STNS-600 nanorods exhibit more uniform dispersion and superior electrocatalytic performance for methanol electrooxidation. The main reason seems to be that both of them are multiphase composites with a large number of phase interfaces and crystal defects, which is conducive to the deposition of Pt nanoparticles. The uniform dispersion of Pt nanoparticles plays an essential role in the electrochemical performance of catalysts. In addition, the presence of the “anatase TiO2” phase in both of them can further enhance the electrochemical performance due to the metal–support interaction. Moreover, compared to commercial Pt/C, the Pt/C-STNS-600 catalyst exhibits higher electrochemical activity and stability, suggesting that superior catalysts can be developed by designing the structure and composition of the supports.

 Please wait while we load your content...
Please wait while we load your content...