Significantly improved photovoltaic performance of the triangular-spiral TPA(DPP–PN)3 by appending planar phenanthrene units into the molecular terminals†
Abstract
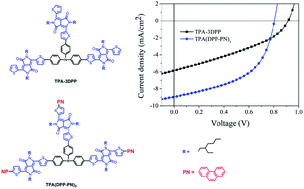
A novel triangular-spiral conjugation molecule of TPA(DPP–PN)3 using triphenylamine (TPA) as the donor core, diketopyrrolopyrrole (DPP) as the acceptor arm and phenanthrene (PN) as the planar arene terminal, as well as its counterpart of TPA–3DPP without the PN terminal, were prepared. Their UV-vis absorption, electrochemistry and thermal stability, as well as hole mobility were investigated. Significantly red-shifted UV-vis absorption profiles were observed for TPA(DPP–PN)3 instead of TPA–3DPP in solution and solid state. A hole mobility of 1.67 × 10−4 cm2 V−1 s−1 was obtained for the TPA(DPP–PN)3/PC71BM blended film, which is 2.1 times higher than that of the TPA–3DPP/PC71BM blended film. Furthermore, the TPA(DPP–PN)3/PC71BM-based organic solar cells presented better photovoltaic property with a maximum power conversion efficiency of 3.67%, which is 1.9 times higher than that of TPA–3DPP/PC71BM-based devices. The results confirm that appending planar PN terminals to TPA–3DPP with a triangular-spiral shape is an efficient approach to improve the photovoltaic performance of its resulting molecules.

 Please wait while we load your content...
Please wait while we load your content...