Efficient nitrogen-doping and structural control of hierarchical carbons using unconventional precursors in the form of deep eutectic solvents†
Abstract
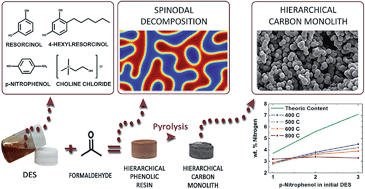
Since the seminal work by Pekala in 1989, polycondensation of phenol derivatives with formaldehyde and subsequent carbonization has been one of the most used procedures for preparation of porous carbons. Nitrogen-doped carbons have also been obtained through this approach only by using nitrogen-rich precursors. The list of the most commonly used nitrogen-rich precursors includes melamine, urea, 3-hydroxypyridine, 3-aminophenol and lysine, and despite a few of them can be used in a single fashion, they typically need to be co-condensed with a second precursor. Nitrogen-rich precursors different from these ones have been used rarely because their molecular structure does not favor the nucleophilic substitution through which polycondensation takes place – e.g. p-nitrophenol. This is by no means a trivial issue because, on the one hand, these precursors cannot form a cross-linked network by themselves, and on the other hand, it may be difficult to encompass their different reaction kinetics when combined with more reactive precursors. This is also the situation for other precursors with an amphiphilic molecular structure that could be of interest to control the structure of the resulting porous carbons – e.g. 4-hexylresorcinol. In this work, we have used deep eutectic solvents composed of resorcinol, 4-hexylresorcinol, p-nitrophenol and choline chloride for the preparation of nitrogen-doped carbon monoliths with a hierarchical porous structure. Carbon conversions ranged from 64 to 50% – depending on the carbonization temperature – despite using three different carbon precursors for co-condensation and two of them were uncommon. The nitrogen content ranged from 4.9 to 3.0 wt%, revealing an excellent nitrogen-doping efficiency for p-nitrophenol when used in the form of DES. Finally, the use of 4-hexylresorcinol controlled the formation of a narrow microporosity that, in combination with the nitrogen functionalities, provided a remarkable CO2-sorption capability to the resulting carbons.

 Please wait while we load your content...
Please wait while we load your content...