One-step synthesis of multi-walled carbon nanotubes/ultra-thin Ni(OH)2 nanoplate composite as efficient catalysts for water oxidation†
Abstract
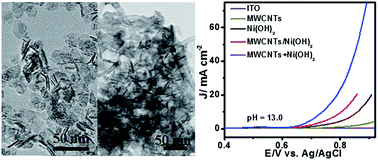
We report a novel approach to synthesize ultra-thin β-Ni(OH)2 nanoplates with a thickness of 1.5–3.0 nm and their composites with multi-walled carbon nanotubes (MWCNTs) by one-step hydrothermal in the absence of surfactants. Ultra-thin β-Ni(OH)2 nanoplates have a large surface area of 139.0 m2 g−1, associated with more exposed surface Ni species, and exhibit better catalytic activity for oxygen evolution reaction (OER) than that of thick β-Ni(OH)2 nanoplates previously reported. Compared to β-Ni(OH)2 nanoplates alone and MWCNTs + Ni(OH)2 nanoplate physical mixture, the composite exhibits much higher electrocatalytic OER activity in terms of low onset overpotential, small Tafel slope, large exchange current density and high OER catalytic current densities at specific applied potentials. The Tafel slope of 87 mV dec−1 for the composite in pH 13 KOH is much smaller than that of β-Ni(OH)2 nanoplates (165 mV dec−1) and their physical mixture (140 mV dec−1). The enhanced catalytic activity of the MWCNTs/Ni(OH)2 composite could be ascribed to the synergic interface of MWCNTs and ultra-thin β-Ni(OH)2 nanoplates for improved conductivity, efficient chemical transfer and high oxidation state of Ni species in the composite electrodes by introducing MWCNTs. No obvious degradation of the OER catalytic current density of the composite electrode over a period of six hours was observed.

 Please wait while we load your content...
Please wait while we load your content...