Templateless electrogeneration of polypyrrole nanostructures: impact of the anionic composition and pH of the monomer solution†
Abstract
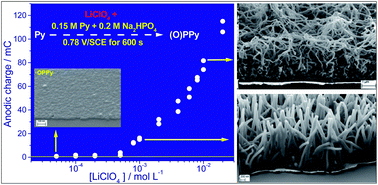
Different superhydrophilic polypyrrole nanostructures can be electrosynthezised in the presence of anions of weak acid (monohydrogenophosphate) and non-acidic anions (perchlorate) without the need for templates. Actually the type of nanostructures formed depends both on the concentration of anions at the electrode and on the interfacial pH. Depending on the anion composition of the pyrrole aqueous solution the film electrogenerated under a given applied potential is either a very thin membrane (10–20 nm) consisting of overoxidized polypyrrole or a tridimensional film with oriented nanowire array or a network of more or less interconnected nanofibers. The formation of such nanostructures is explained by a side reaction which is water oxidation. Since this reaction is pH-dependent, the pH of the pyrrole solution is one of the key parameter for the synthesis of such nanostructures. The reaction mechanism is discussed and compared to those proposed in the literature for nanofiber network electrosynthesis. Actually in the monomer solution, the role of the anions of weak acid is twofold. On the one hand they allow to limit the decrease of the interfacial pH during pyrrole oxidation and on the other hand to decrease the interfacial anion concentration, so that water oxidation takes place with formation of hydroxyl radicals and dioxygen nanobubbles.

 Please wait while we load your content...
Please wait while we load your content...