Nanocrystalline-Li2FeSiO4 synthesized by carbon frameworks as an advanced cathode material for Li-ion batteries
Abstract
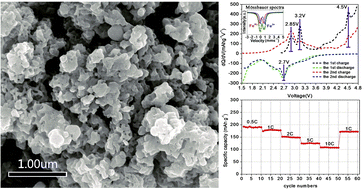
The P21/n structured nanocrystalline-Li2FeSiO4 is prepared by a confinement effect of three-dimensional conductive carbon frameworks, which are formed through a chelating reaction and subsequent pyrolysis. As a benefit of enhanced electronic conductivity by carbon frameworks and Li-ion diffusion kinetics by nanocrystalline-Li2FeSiO4 architectures, the novel nanocomposite shows a 1.28 Li-ion storage capacity (211.3 mA h g−1) at 0.1 C, corresponding to two successive steps of oxidation and reduction of Fe2+/Fe3+/Fe4+. Furthermore, the discharge capacity is 189.8, 175.6, 148.9, 125.7 and 106.6 mA h g−1 at a variable rate of 0.5, 1, 2, 5 and 10 C, respectively, and then easily returns to 175 mA h g−1 at 1 C. It is a surprise that the initial capacity is 90.9 mA h g−1 at 10 C, and 97.7% is retained after 1000 cycles. Thus, we believe that the nanocrystalline-Li2FeSiO4 with carbon frameworks, possessing high-capacity and high-rate performance, is a promising next-generation cathode material for high-power lithium-ion batteries.

 Please wait while we load your content...
Please wait while we load your content...