Influence of cyclic versus acyclic oxygen-containing electron donor ancillary ligands on the photocurrent, photovoltage and photostability for high efficiency dye-sensitized solar cells†
Abstract
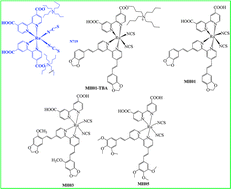
Three novel heteroleptic amphiphilic polypyridyl Ru-complexes, MH01, MH03, and MH05, with oxygen-containing-electron-donor stilbazole-based ancillary ligands were synthesized to study the influence of the cyclic-electron-donor (MH01), the presence of the cyclic electron donor coupled to acyclic electron-donor auxochromes (MH03) ortho to the CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH bridge of stilbazole, and the presence of only acyclic electron-donor methoxy group (MH05) on molar extinction coefficient, light harvesting efficiency (LHE), ground and excited state oxidation potentials, and photovoltaic performance for DSSCs. Although MH05 has three electron donor methoxy groups, it achieved the lowest molar extinction coefficient of 18 250 M−1 cm−1 and exhibited the lowest photocurrent. The highest photocurrent density (JSC) was observed for the longest interatomic distance between the CH
CH bridge of stilbazole, and the presence of only acyclic electron-donor methoxy group (MH05) on molar extinction coefficient, light harvesting efficiency (LHE), ground and excited state oxidation potentials, and photovoltaic performance for DSSCs. Although MH05 has three electron donor methoxy groups, it achieved the lowest molar extinction coefficient of 18 250 M−1 cm−1 and exhibited the lowest photocurrent. The highest photocurrent density (JSC) was observed for the longest interatomic distance between the CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH bridge of the stilbazole moiety and cyclic-electron-donor auxochromes (MH01). It was also shown that while incorporation of acyclic electron-donor auxochromes ortho to the CH
CH bridge of the stilbazole moiety and cyclic-electron-donor auxochromes (MH01). It was also shown that while incorporation of acyclic electron-donor auxochromes ortho to the CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH bridge (MH03) has little effect on the ground and excited state oxidation potentials, λmax of the low energy MLCT, and molar absorptivity, the lowest photovoltage and %η were observed. When compared under the same experimental device conditions using 0.3 M tert-butylpyridine (TBP), only MH01-TBA achieved 18% more in JSC and 8.6% greater in η than the benchmark dye N719. To probe the interrelationship among the cyclic-vs.-acyclic oxygen-containing electron donors of the ancillary ligands, photocurrent and photovoltage of these dyes, the equilibrium molecular geometries of the ancillary ligands were calculated using DFT. The HOMO distribution on cyclic-vs.-acyclic electron donors and the position of OMe in the ancillary ligands rationalized the fundamental science behind the photovoltaic performance and photostability of these dyes.
CH bridge (MH03) has little effect on the ground and excited state oxidation potentials, λmax of the low energy MLCT, and molar absorptivity, the lowest photovoltage and %η were observed. When compared under the same experimental device conditions using 0.3 M tert-butylpyridine (TBP), only MH01-TBA achieved 18% more in JSC and 8.6% greater in η than the benchmark dye N719. To probe the interrelationship among the cyclic-vs.-acyclic oxygen-containing electron donors of the ancillary ligands, photocurrent and photovoltage of these dyes, the equilibrium molecular geometries of the ancillary ligands were calculated using DFT. The HOMO distribution on cyclic-vs.-acyclic electron donors and the position of OMe in the ancillary ligands rationalized the fundamental science behind the photovoltaic performance and photostability of these dyes.

 Please wait while we load your content...
Please wait while we load your content...