A one-step delamination procedure to form single sheet iron(iii)-(oxy)hydroxides†
Abstract
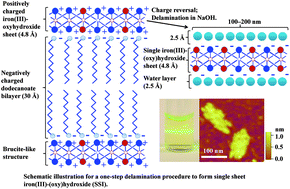
The dispersion of a layered iron(III)-(oxy)hydroxide intercalated with dodecanoate (oxGRC12, Fe3IIIO2.18(OH)3.13(C12H23O2)0.56(SO4)0.47; derived from the corresponding layered iron(II)–iron(III)-hydroxide) in 0.1 M sodium hydroxide results in delamination of oxGRC12 with formation of separate planar layers of iron(III)-(oxy)hydroxides, here termed single sheet iron-(oxy)hydroxides (SSI). Delamination is confirmed by powder X-ray diffraction while Fourier transform infrared spectroscopy reveals the removal of dodecanoate from the parent compound. Atomic force microscopy shows that SSI has a thickness of 1 nm and the lateral size ranging from 100 to 200 nm. The observed thickness of SSI is double the thickness of the iron(III)-(oxy)hydroxide layers (0.48 nm) which is attributed to water layers adsorbed on both sides of SSI. The SSI shows a high tendency to aggregate. High resolution transmission electron microscopy and selected area electron diffraction confirm that SSI inherits the in-plane crystal structure from its parent layered compound. Both the hyperfine parameters and the X-ray absorption spectrum show a similar local iron coordination in SSI before and after delamination. The extended X-ray absorption fine structure spectra also show that the FeO6 octahedron has expanded to some extent (1–2%) after delamination. O1s X-ray photoelectron spectra of SSI reveal extensive deprotonation of hydroxyl groups in SSI, reflecting a layer charge reversal during delamination. To the best of our knowledge, this is the first report on planar 2D nanosheets of iron-(oxy)hydroxide with the thickness of 1 nm.

 Please wait while we load your content...
Please wait while we load your content...