Catalytic and synergistic effects on thermal stability and combustion behavior of polypropylene: influence of maleic anhydride grafted polypropylene stabilized cobalt nanoparticles
Abstract
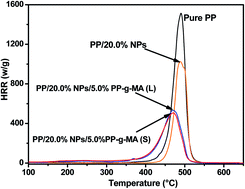
A novel function of polypropylene grafted with maleic anhydride (PP-g-MA) as synergistic additives to effectively reduce the combustion rates of polypropylene (PP) polymer nanocomposites (PNCs) reinforced with in situ synthesized cobalt (Co)–cobalt oxide core–shell nanoparticles (NPs) was reported. Two PP-g-MAs with molecular weights of ∼800 and 2500 g mol−1 have been chosen to demonstrate this function. The combustion behaviors and thermal stability of the PP/Co PNCs were evaluated by micro-scale combustion calorimetry (MCC) and thermal gravimetric analysis (TGA). MCC result illustrated that the combustion behaviors of PP were altered by a reduction of ∼32% in the peak heat release rate (PHRR) with the incorporation of 20.0 wt% Co NPs; more importantly, further reduction in PHRR more than 50% was observed by adding only 5.0 wt% non-flame retardant PP-g-MA into PP/20.0 wt% Co system. This flame retardancy was elucidated through combining MCC with a fast TGA thermal degradation technique. The change of PP thermal degradation pathway by the synergistic and catalytic effects of PP-g-MA and Co NPs in the PP matrix was found to be responsible for the decreased combustion rates of PP. Strong interactions between PP and Co NPs formed by the PP-g-MA was further confirmed by attenuated total reflection-Fourier transform infrared spectroscopy (ATR-FTIR), which favor the decrease of interfacial tension. Differential scanning calorimetry (DSC) and melt rheometry also demonstrated the surfactant molecular weight dependent melting and crystalline properties, and the melt rheological behaviors of the PP matrix.

 Please wait while we load your content...
Please wait while we load your content...