Aqueous self-assembly of hydrophobic macromolecules with adjustable rigidity of the backbone†
Abstract
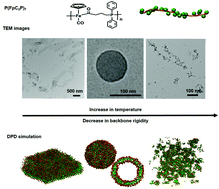
P(FpC3P) (Fp: CpFe(CO)2; C3P: propyl diphenyl phosphine) has a helical backbone, resulting from piano stool metal coordination geometry, which is rigid with intramolecular aromatic interaction of the phenyl groups. The macromolecule is hydrophobic, but the polarized CO groups can interact with water for aqueous self-assembly. The stiffness of P(FpC3P), which is adjustable by temperature, is an important factor influencing the morphologies of kinetically trapped assemblies. P(FpC3P)7 self-assembles in DMSO/water (10/90 by volume) into lamellae at 25 °C, vesicles at 40 °C and irregular aggregates at higher temperatures (60 and 70 °C). The colloidal stability decreases in the order of lamellae, vesicles and irregular aggregates. Dissipative particle dynamics (DPD) simulation reveals the same temperature-dependent self-assembled morphologies with an interior of hydrophobic aromatic groups covered with the metal coordination units. The rigid backbone at 25 °C accounts for the formation of the layered morphology, while the reduced rigidity of the same P(FpC3P)7 at 40 °C curves up the lamellae into vesicles. At a higher temperature (60 or 70 °C), P(FpC3P)7 behaves as a random coil without obvious amphiphilic segregation, resulting in irregular aggregates. The stiffness is, therefore, a crucial factor for the aqueous assembly of macromolecules without obvious amphiphilic segregation, which is reminiscent of the solution behavior observed for many hydrophobic biological macromolecules such as proteins.


 Please wait while we load your content...
Please wait while we load your content...