Universal scaling of crowding-induced DNA mobility is coupled with topology-dependent molecular compaction and elongation
Abstract
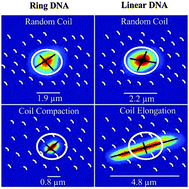
Using single-molecule fluorescence microscopy and particle-tracking techniques, we elucidate the role DNA topology plays in the diffusion and conformational dynamics of crowded DNA molecules. We focus on large (115 kbp), double-stranded ring and linear DNA crowded by varying concentrations (0–40%) of dextran (10, 500 kDa) that mimic cellular conditions. By tracking the center-of-mass and measuring the lengths of the major and minor axes of single DNA molecules, we characterize both DNA mobility reduction as well as crowding-induced conformational changes (from random spherical coils). We reveal novel topology-dependent conformations, with single ring molecules undergoing compaction to ordered spherical configurations ∼20% smaller than dilute random coils, while linear DNA elongates by ∼2-fold. Surprisingly, these highly different conformations result in nearly identical exponential mobility reduction dependent solely on crowder volume fraction Φ, revealing a universal critical crowding concentration of Φc ≅ 2.3. Beyond Φc DNA exhibits topology-independent conformational relaxation dynamics despite highly distinct topology-driven conformations. Our collective results reveal that topology-dependent conformational changes, unique to crowded environments, enable DNA to overcome the classically expected mobility reduction that high-viscosity crowded environments impose. Such coupled universal dynamics suggest a mechanism for DNA to maintain sufficient mobility required for wide-ranging biological processes despite severe cellular crowding.

 Please wait while we load your content...
Please wait while we load your content...