Mechanism of two-dimensional crystal formation from soft microgel particles†
Abstract
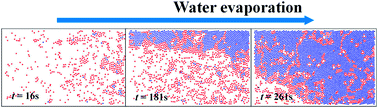
The mechanism of two-dimensional (2D) crystal formation from an air-drying suspension of soft microgel particles is investigated via in situ confocal laser scanning microscopy (CLSM). According to the time-resolved radial distribution function g(r), the process can be divided into two separate stages, i.e., nucleation and growth. During the air-drying process, the soft poly(N-isopropylacrylamide-co-methacrylic acid) (P(NIPAM-co-MAA)) spherical microgel particles first deform into a disk-like shape as the thickness of the concave water layer becomes comparable to their diameter. Subsequently, the particles are driven together by attractive capillary forces to initiate the nucleation process. Once the critical nuclei size is reached, the 2D nuclei can then grow into a crystal. Subsequently, during the crystal growth process, convective water flow compels free microgel particles from the liquid phase to the crystal growth front. While trapped in the growth front, the particles rearrange themselves into crystal structures. The size of 2D crystals increases quadratically with time. Ideally, these findings will promote more experimental and theoretical work aimed at elucidating the 2D fluid-to-solid transition of soft microgel particles, as well as 2D soft disk crystal growth in condensed matter physics.

 Please wait while we load your content...
Please wait while we load your content...