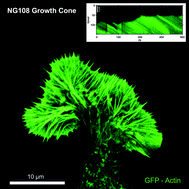
The lamellipodium, a thin veil-like structure at the leading edge of motile cells, is fundamental for cell migration and growth. Orchestrated activities of membrane components and an underlying biopolymer film result in a controlled movement of the whole system. Dynamics in two-dimensional cell motility are primarily driven by the actively moving protein film in the lamellipodium. Polymerization of actin filaments at the leading edge, back-transport of the actin network due to myosin motor activity, depolymerization in the back, and diffusive transport of actin monomers to the front control these dynamics. The same molecular prerequisites for lamellipodial motion are found in most eukaryotic cells and can function independently of the cell body. Here we show that lamellipodial dynamics differ strongly in different cell types according to their function. Path finding neuronal growth cones display strong stochastic fluctuations, wound healing fibroblasts that locally migrate in tissues exhibit reduced fluctuations while fish keratocytes move highly persistently. Nevertheless, experimental analysis and computer simulations show that changes in the parameters for actin polymerization and retrograde actin transport alone are sufficient for the cell to utilize the same, highly adaptive machinery to display this rich variety of behaviors.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...