The hydrophobic effect as a driving force for charge inversion in colloids
Abstract
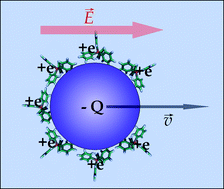
A new mechanism for charge inversion of colloids in electrolyte solutions based on the hydrophobic effect is presented. We show that this mechanism is able to induce charge inversion at low concentrations (∼mM) of monovalent ions containing hydrophobic functional groups and present for the first time experimental evidence of this prediction. We provide a simple mean field theoretical model that successfully describes the experimental data up to charge inversion. Saturation effects appearing at higher concentrations are also discussed.

 Please wait while we load your content...
Please wait while we load your content...