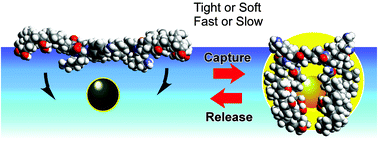
Dynamic aspects of molecular recognition at the air–water interface have been investigated using a monolayer of a steroid cyclophane SC(OH) which consists of the rigid 1,6,20,25-tetraaza[6.1.6.1]paracyclophane ring, connected to four steroid moieties (cholic acid) through flexible L-lysine spacers. An aqueous fluorescent guest (TNS) can be reversibly captured by SC(OH) with a variation in the accompanying fluorescence emission upon compression and expansion of the SC(OH) monolayer. Tight capture, by compression of the monolayer to a high surface pressure, efficiently enhances fluorescence intensity because of suppression of formation of the non-emissive state. On the other hand, rapid motion of the reversible cavity formation by a high rate of compression and expansion of the monolayer results in better reproducibility in the fluorescence change than that obtained under a slower motion, which can be explained by the suppression of unfavorable structural relaxation within the monolayer structures. This result has connotations for the development of novel molecular devices and machines that operate through mechanically driven molecular recognition.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...