A two-dimensional arsenene/g-C3N4 van der Waals heterostructure: a highly efficient photocatalyst for water splitting
Abstract
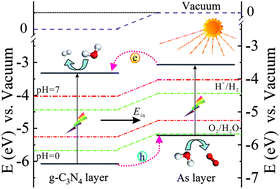
Producing hydrogen through photocatalytic water splitting is a feasible and efficient route for resolving the problems associated with energy and the environment, and great efforts have been devoted to improving solar-to-hydrogen (STH) efficiency. Here, we study an arsenene/g-C3N4 van der Waals heterostructure as a possible photocatalyst for water splitting. Using first principles calculations, we find that the heterostructure is an indirect-gap semiconductor with a type-II band alignment and low exciton binding energy, facilitating the effective separation of photogenerated electrons and holes. Comparing the band edge positions with the redox potentials of water demonstrates that the heterostructure is a potential photocatalyst for water splitting in a neutral environment. Meanwhile, the heterostructure shows significantly enhanced optical absorption beyond the constituent monolayers in both visible and ultraviolet regions. Its STH efficiency limit can reach up to 15.8%, which is quite promising for commercial applications. Thermodynamic analyses for redox reactions suggest facile hydrogen generation on the heterostructure. These extraordinary properties enable the arsenene/g-C3N4 heterostructure to be a promising water-splitting photocatalyst.


 Please wait while we load your content...
Please wait while we load your content...