Combining battery-like and pseudocapacitive charge storage in 3D MnOx@carbon electrode architectures for zinc-ion cells†
Abstract
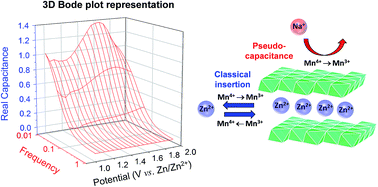
We demonstrate that electrodes comprising nanoscale, birnessite-type manganese oxide affixed to carbon nanofoam paper (MnOx@CNF) exhibit two distinct charge-storage mechanisms—battery-like Zn2+ insertion/de-insertion and pseudocapacitance—when electrochemically cycled in aqueous electrolytes that include both Na+ and Zn2+ salts. When the mixed-electrolyte composition is 0.75 M Na2SO4 + 0.25 M ZnSO4 (i.e., “6[Na+] : 1[Zn2+]”), the MnOx@CNF electrode delivers high specific capacity at low rates, approaching theoretical capacity for Zn2+ insertion/de-insertion at MnOx. At high rates (>10C) the Na+-supported pseudocapacitance mechanism maintains charge-storage capacity well above that observed with electrolytes that contain only ZnSO4. Impedance analysis was performed to discriminate between these distinct charge-storage mechanisms by visualizing the frequency- and potential-dependent capacitance as 3D Bode plots. In the 6[Na+] : 1[Zn2+] electrolyte, the potential-independent pseudocapacitance is augmented by reversible Zn2+-based redox processes between 1.4 and 1.8 V vs. Zn/Zn2+. Galvanostatic testing with two-electrode zinc-ion cells that pair MnOx@CNF with a zinc foil negative electrode proves the practical performance advantages of combining pseudocapacitance and Zn2+-insertion mechanisms: higher energy efficiency and greater specific power in the 6[Na+] : 1[Zn2+] electrolyte compared to 1 M ZnSO4.


 Please wait while we load your content...
Please wait while we load your content...