Evaluating spinel ferrites MFe2O4 (M = Cu, Mg, Zn) as photoanodes for solar water oxidation: prospects and limitations†
Abstract
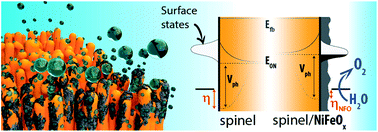
The search for ideal semiconductors for photoelectrochemical solar fuel conversion has recently recognized the spinel ferrites as promising candidates due to their optoelectronic tunability together with superb chemical stability. However, a systematic understanding of the main material factors limiting their performance is currently lacking. Herein, nanostructured thin-film electrodes of three representative spinels, namely CuFe2O4 (CFO), MgFe2O4 (MFO) and ZnFe2O4 (ZFO), are prepared by a solution-based approach and their photoelectrochemical (PEC) properties are comprehensively characterized. Annealing post-treatments together with the deposition of NiFeOx overlayers are found to improve the native n-type response, although a dominant bulk recombination (especially in MFO) limits the saturation photocurrents (below 0.4 mA cm−2 at 1.23 V vs. RHE). Likewise, prominent Fermi level pinning due to surface states at around 0.9 V vs. RHE in all cases appears to limit the photovoltage (to ca. 300 mV). Rapid-scan voltammetry is used to gain insight into the surface states and the operation of the overlayer. Interestingly, the NiFeOx is ineffective at mitigating Fermi level pinning, but clearly participates as an electrocatalyst to improve the overall performance. Generally, these results evidence the potential and current intrinsic limitations of the spinel ferrites—establishing a roadmap for the optimization of these materials as photoanodes for solar water oxidation.
- This article is part of the themed collection: 3rd International Solar Fuels Conference and International Conference on Artificial Photosynthesis


 Please wait while we load your content...
Please wait while we load your content...